Carathéodory's work was taken up by Max Born, and it is treated in a few monographs. His axiom replaced the earlier Kelvin-Planck and the equivalent Clausius formulation of the second law and did not need Carnot engines. His theory was based on Pfaffian differential equations. Not satisfied with the engineering type of argument, the mathematician Constantin Carathéodory gave in 1909 a new axiomatic formulation of entropy and the second law of thermodynamics. For a system of about 10 23 particles, Ω is on the order of 10 10 23, that is the entropy is on the order of 10 23× k B ≈ R, the molar gas constant. The number Ω is the multiplicity of the macrostate for an isolated system, where the macrostate is of definite energy, Ω is its degeneracy. Ω is the number of different wave functions ("microstates") of the system belonging to the system's "macrostate" (thermodynamic state). In the statistical approach the entropy of an isolated (constant energy) system is k B log Ω, where k B is Boltzmann's constant and the function log stands for the natural (base e) logarithm. 
The quantum statistical point of view, too, will be reviewed in the present article. Boltzmann's definition of entropy was furthered by John von Neumann to a quantum statistical definition. 
In 1877 Ludwig Boltzmann gave a definition of entropy in the context of the kinetic gas theory, a branch of physics that developed into statistical thermodynamics. The second law states that the entropy of an isolated system increases in spontaneous (natural) processes leading from one state to another, whereas the first law states that the internal energy of the system is conserved. In this approach, entropy is the amount of heat (per degree kelvin) gained or lost by a thermodynamic system that makes a transition from one state to another. The "engineering" manner-by an engine-of introducing entropy will be discussed below. Carnot's work foreshadowed the second law of thermodynamics. The traditional way of introducing entropy is by means of a Carnot engine, an abstract engine conceived of by Sadi Carnot in 1824 as an idealization of a steam engine. The state variable "entropy" was introduced by Rudolf Clausius in 1865, see the inset for his text, when he gave a mathematical formulation of the second law of thermodynamics. I have deliberately constructed the word entropy to resemble as much as possible the word energy, since both quantities to be named by these words are so closely related in their physical meaning that a certain similarity in their names seems appropriate to me. 
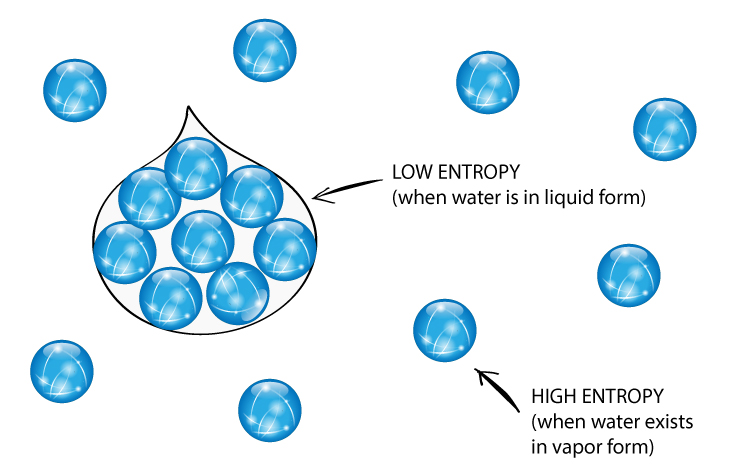
As I deem it better to derive the names of such quantities - that are so important for science - from the antique languages, so that they can be used without modification in all modern languages, I propose to call the quantity S the entropy of the body, after the Greek word for transformation, ἡ τροπή. Hence a macroscopic sample of a gas occupies all of the space available to it, simply because this is the most probable arrangement.Įntropy depends not only on the number of atoms or molecules and the volume of available space, but also their freedom of motion, which corresponds to temperature and state of matter.Translation: Searching for a descriptive name for S, one could - like it is said of the quantity U that it is the heat and work content of the body - say of the quantity S that it is the transformation content of the body. The probability of arrangements with essentially equal numbers of molecules in each bulb is quite high, however, because there are many equivalent microstates in which the molecules are distributed equally. Although nothing prevents the molecules in the gas sample from occupying only one of the two bulbs, that particular arrangement is so improbable that it is never actually observed. Instead of four molecules of gas, what if we had one mole of gas, or 6.022 × 10 23 molecules in the two-bulb apparatus? If we allow the sample of gas to expand spontaneously in the two containers, the probability of finding all 6.022 × 10 23 molecules in one container and none in the other at any given time is extremely small, effectively zero. \( \newcommand\): The Possible Microstates for a Sample of Four Gas Molecules in Two Bulbs of Equal Volume 
0 Comments
Leave a Reply. |
Details
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
